Inhibitors of TRIP8b for Novel Mood Disorder Therapeutics
SHORT DESCRIPTION
Novel inhibitors that disrupt TRIP8b-HCN binding to offer an alternative antidepressant mechanism for treatment of mood disorders.
INVENTORS
- Gary Schiltz*
- Weinberg College of Arts and Sciences, Department of Chemistry
- Dane Chetkovich
- Ye Han
- Iredia Iyamu
- Kyle Lyman
* Principal Investigator |
NU Tech ID NU 2021-223
IP STATUS
US Patent Pending (18/330,874)
DEVELOPMENT STAGE
TRL-3 - Experimental Proof-of-Concept: Biochemical and cell-based assays have validated the compound's ability to disrupt TRIP8b-HCN interactions.
|
BACKGROUND
Major Depressive Disorder (MDD) is one of the most prevalent and disabling conditions in medicine, affecting an estimated 17% of the U.S. population over a lifetime and ranking among the leading causes of years lived with disability worldwide. In any given year, about 21 million American adults experience at least one major depressive episode, characterized by persistent low mood, anhedonia, cognitive impairment, and functional decline, with enormous personal, societal, and economic costs. Current treatment relies largely on monoaminergic agents that increase synaptic serotonin, norepinephrine, and dopamine, yet many patients experience delayed onset, partial response, residual cognitive and functional deficits, or dose‑limiting side effects such as sexual dysfunction, weight gain, and sleep disturbance, leaving a significant proportion symptomatic or treatment‑resistant. This creates a clear need for mechanistically distinct antidepressants that can help non‑responders, act more rapidly, and offer improved tolerability. Emerging data implicate hyperpolarization‑activated cyclic nucleotide‑gated (HCN) channels and their auxiliary subunit TRIP8b in regulating neuronal excitability and mood circuitry, and preclinical studies show that genetic deletion or pharmacological antagonism of HCN channels or TRIP8b produces robust antidepressant‑like effects. Inhibitors that disrupt TRIP8b–HCN interactions therefore represent a compelling opportunity to move beyond monoamine‑based mechanisms, overcome key limitations of current treatments, and directly address this unmet need.
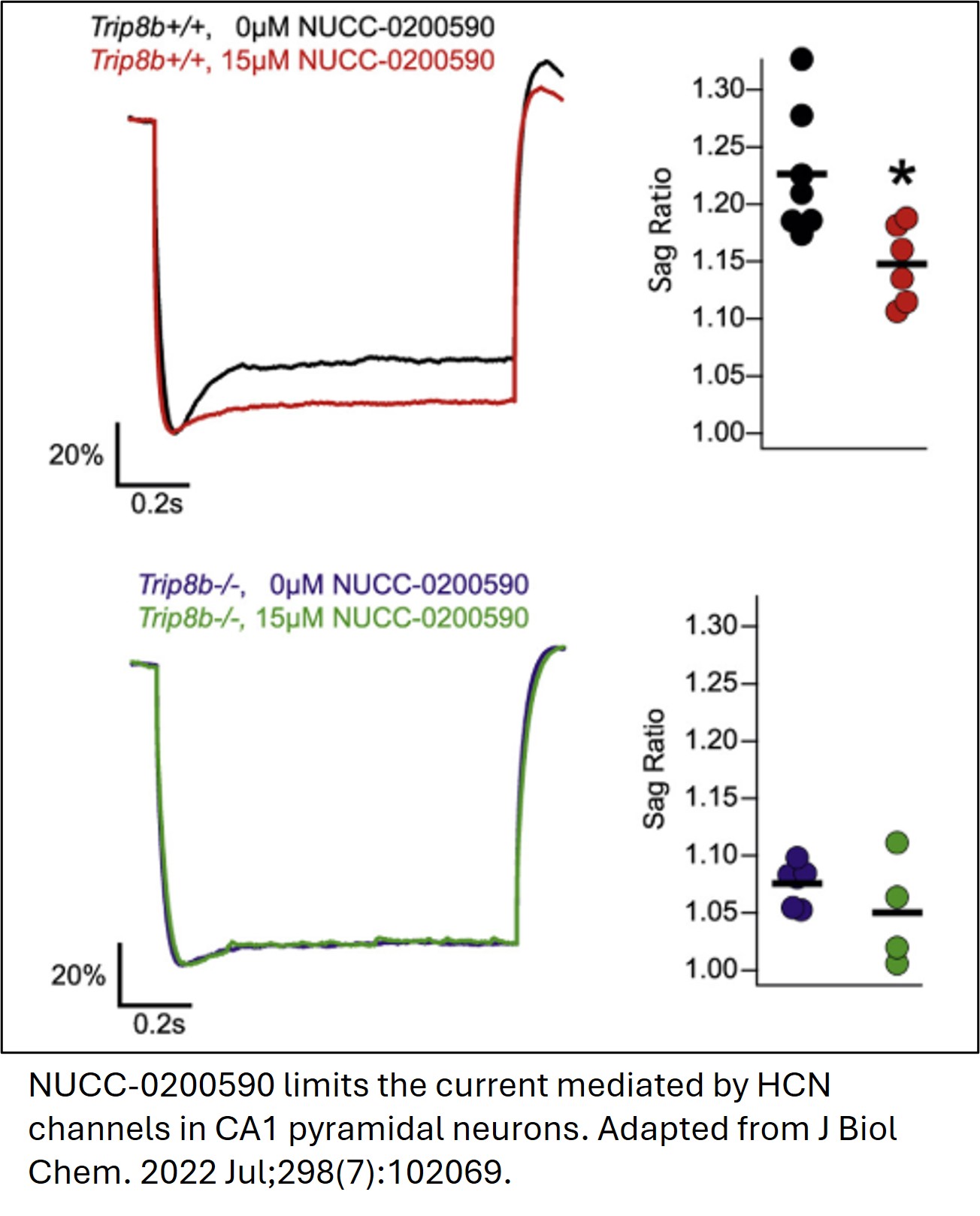 ABSTRACT
ABSTRACT
Northwestern University researchers have developed a series of novel small-molecule compounds that selectively disrupt the binding between TRIP8b and HCN channels to modulate HCN channel trafficking and function in neurons. Lead compound NUCC-0200590 and a broad library of structurally related analogs were identified through virtual high-throughput screening and subsequent biochemical, biophysical, and cell-based assays, as well as ex vivo mouse hippocampal tissue preparations, confirmed that the compound effectively disrupts TRIP8b-HCN interactions. Representative compounds achieve IC50 values in the low micromolar to sub‑micromolar range, while maintaining acceptable cell viability across relevant concentration ranges. This work provides a promising template for developing innovative first-in-class antidepressant therapies with a distinct mechanism of action for for Major Depressive Disorder (MDD) and related mood disorders.
APPLICATIONS
- Treatment of Major Depressive Disorder (MDD), including patients with inadequate response or intolerance to existing monoaminergic antidepressants, by oral or parenteral administration
- Treatment for other mood disorders including dysthymia, bipolar depression, mood disorders related to a comorbid medical condition, and substance-induced mood disorders.
- Research tool compounds for probing TRIP8b–HCN biology and dissecting the contribution of hippocampal HCN channel regulation to affective behavior.
- Platform for rational drug discovery: the disclosed chemical scaffold and structure–activity relationship data provide a medicinal chemistry foundation for developing optimized drug candidates with improved potency, selectivity, and pharmacokinetic profiles.
- Potential utility in other neurological conditions where HCN channel dysregulation contributes to pathophysiology, including epilepsy and neuropathic pain.
ADVANTAGES
- Novel mechanism of action: First‑in‑class targeting of the TRIP8b–HCN interaction, providing a unique therapeutic avenue distinct from all currently approved antidepressants.
- Validted target: TRIP8b–HCN interaction is a validated regulator of hippocampal excitability and stress‑induced depressive phenotypes
- Potential for higher efficacy: Mechanistically distinct from current antidepressants, creating opportunity in treatment‑resistant populations and enabling rational combinations without overlapping primary targets
- Strong preclinical validation: Lead compound NUCC‑0200590 and related analogs been characterized across orthogonal biochemical, biophysical, cell-based, and ex vivo assays.
- Broad and growing compound library offers flexibility to optimize potency, selectivity, CNS exposure, and safety
- Versatile formulation potential: Small‑molecule scaffolds amenable to scalable synthesis and formulation into standard oral and parenteral dosage forms
- Broad applicability: Enables further drug development for a range of neurological disorders
PUBLICATIONS
 KEYWORDS
KEYWORDS
TRIP8b, HCN channel, antidepressant, mood disorder, small molecule, inhibitor, high-throughput screening, virtual screening, Major Depressive Disorder, neuroscience, neurology, depression
Patent Information:
| Title |
App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |
Categories:
Life Sciences > Therapeutics
Keywords:
Antidepressant
Depression
Mood disorder
Neurologic disease
Neurology
Psychiatric disease
Targeted therapy
Therapeutics