N-desmethyl Tamoxifen and Derivatives for Effective Pain Relief
SHORT DESCRIPTION
A novel state-dependent tamoxifen‑derived small molecule anti-pain medication that selectively blocks peripheral voltage‑gated sodium channels to deliver potent, non‑opioid analgesia in chronic and inflammatory pain.
INVENTORS
- Paul DeCaen*
- Northwestern University Feinberg School of Medicine, Department of Pharmacology
- Thuy Vien*
- Northwestern University Feinberg School of Medicine, Department of Pharmacology
* Principal Investigator |
NU Tech ID NU 2021-099
IP STATUS US Patent Pending (18/054,527)
DEVELOPMENT STAGE
TRL-5 Prototype Validated in Relevant Environment: In vivo rodent models confirm efficacy.
|
BACKGROUND
 Chronic nociceptive and neuropathic pain affects tens of millions of patients annually and remains one of the largest unmet medical needs, driving disability, reduced quality of life, and enormous healthcare and productivity costs. Standard treatments for moderate to severe pain rely heavily on opioids, NSAIDs, and psychotropic agents such as tricyclic antidepressants: opioids are effective but carry high risks of tolerance, dependence, and addiction, while NSAIDs and TCAs offer limited efficacy and are associated with significant cardiovascular and cognitive side effects. Sodium channel blockers like carbamazepine and cannabidiol are used in chronic neuropathic pain (e.g., painful diabetic neuropathy), but they often show low efficacy and limited selectivity for pain‑driving neurons, constraining dose by systemic adverse events. Voltage‑gated sodium channels in sensory neurons are genetically and functionally validated pain targets, and non‑addictive sodium channel inhibition has attracted intense pharmaceutical investment; however, many candidates fail because they lack sufficient potency, state dependence, or safety margin. There is thus a clear unmet need for peripherally acting, non‑opioid analgesics that preferentially inhibit hyperexcitable sensory neurons via novel binding sites on voltage-gated sodium channels while minimizing CNS and off‑target hormonal effects.
Chronic nociceptive and neuropathic pain affects tens of millions of patients annually and remains one of the largest unmet medical needs, driving disability, reduced quality of life, and enormous healthcare and productivity costs. Standard treatments for moderate to severe pain rely heavily on opioids, NSAIDs, and psychotropic agents such as tricyclic antidepressants: opioids are effective but carry high risks of tolerance, dependence, and addiction, while NSAIDs and TCAs offer limited efficacy and are associated with significant cardiovascular and cognitive side effects. Sodium channel blockers like carbamazepine and cannabidiol are used in chronic neuropathic pain (e.g., painful diabetic neuropathy), but they often show low efficacy and limited selectivity for pain‑driving neurons, constraining dose by systemic adverse events. Voltage‑gated sodium channels in sensory neurons are genetically and functionally validated pain targets, and non‑addictive sodium channel inhibition has attracted intense pharmaceutical investment; however, many candidates fail because they lack sufficient potency, state dependence, or safety margin. There is thus a clear unmet need for peripherally acting, non‑opioid analgesics that preferentially inhibit hyperexcitable sensory neurons via novel binding sites on voltage-gated sodium channels while minimizing CNS and off‑target hormonal effects.
ABSTRACT
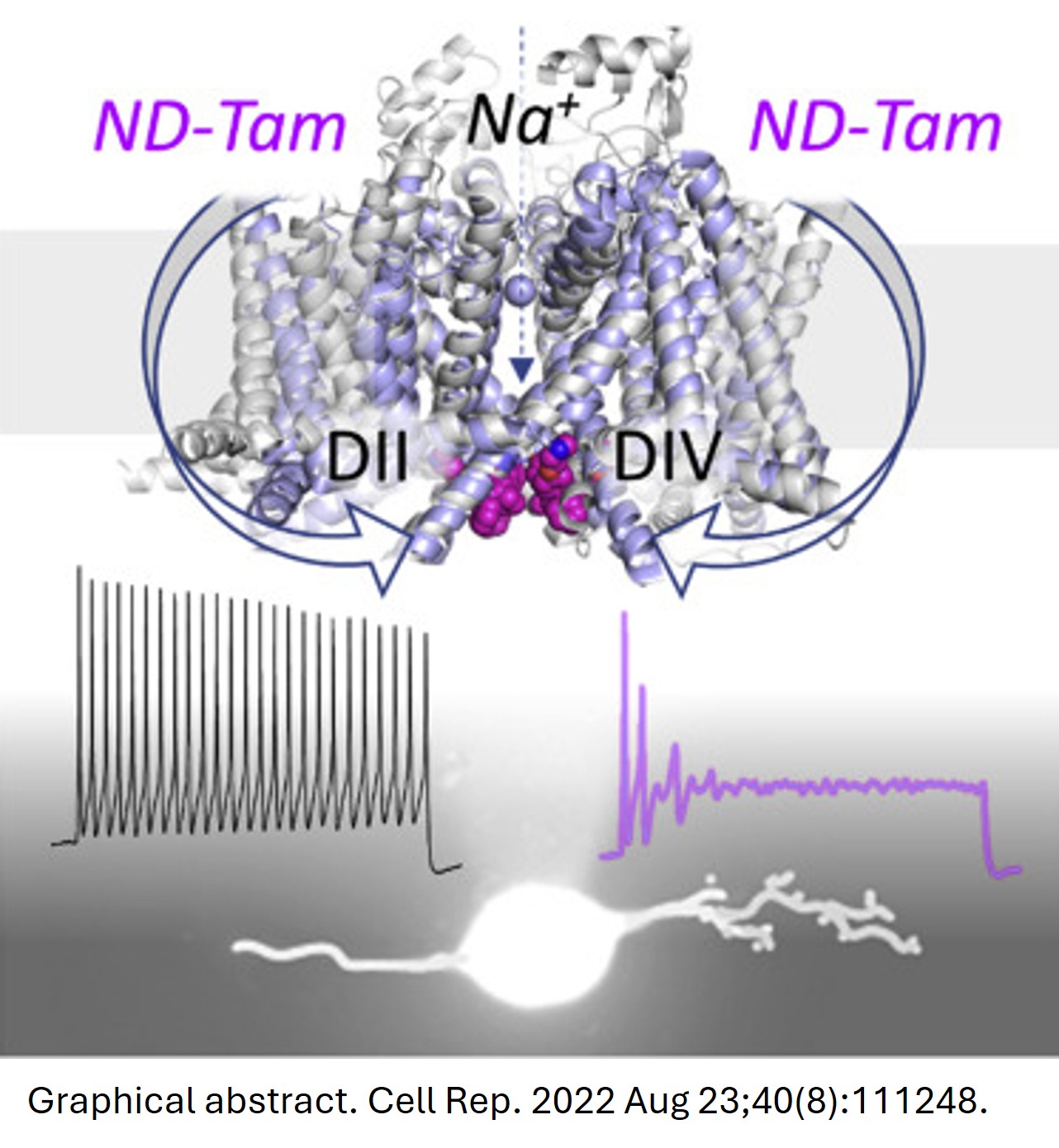
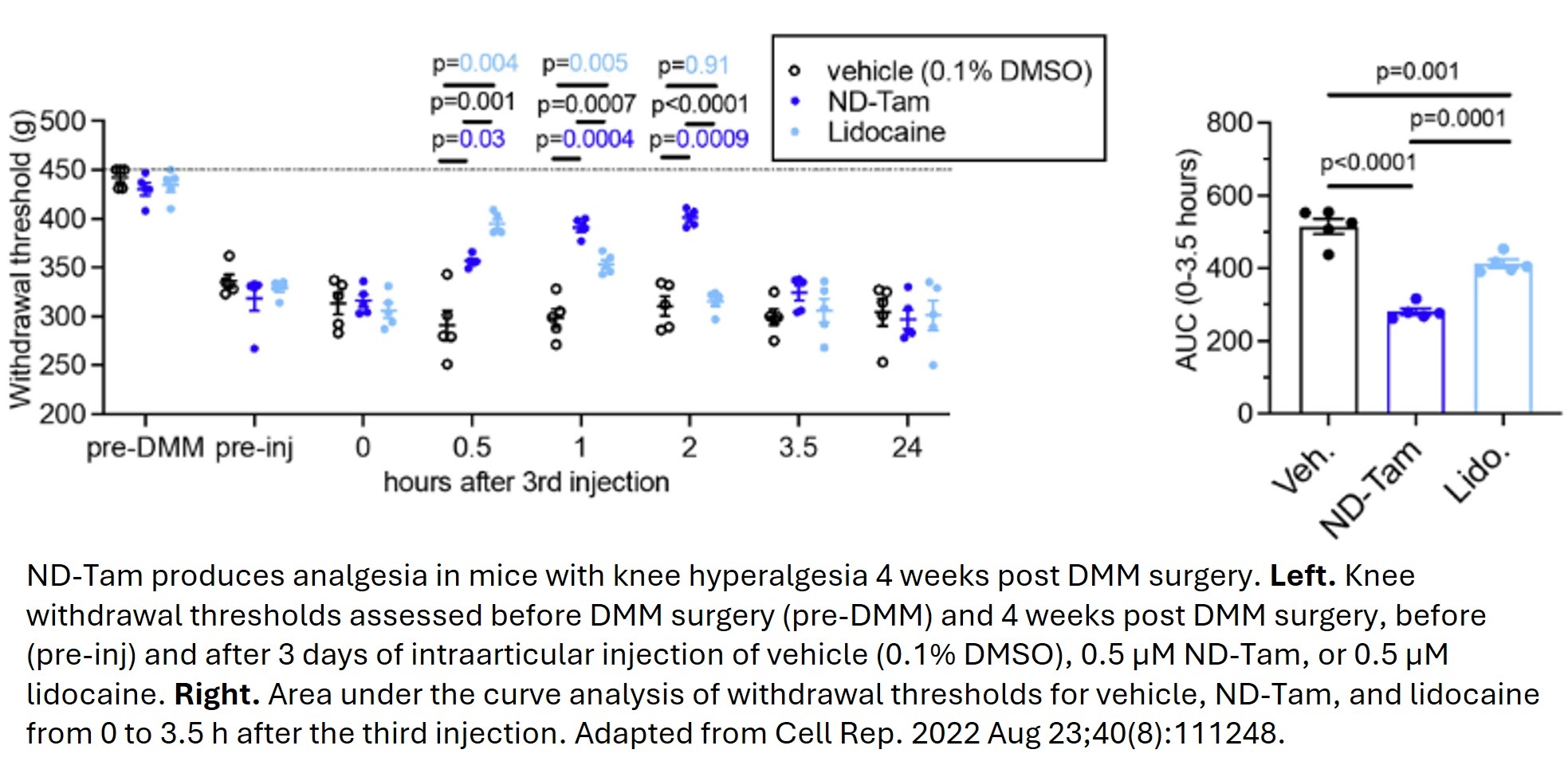 This invention introduces tamoxifen‑derived small molecules, led by N‑desmethyl tamoxifen (ND‑Tam), that bind a newly defined “tamoxifen receptor” site inside voltage‑gated sodium channels in sensory neurons as novel candidates for potent, state‑dependent pain relief. By blocking voltage-gated sodium channels (Nav) more strongly when they are active or inactivated at depolarized potentials, these compounds preferentially silence hyperexcitable nociceptors while sparing normally firing neurons. The inventors demonstrate robust inhibition of sodium currents and action potential firing in vitro in dorsal root ganglion (DRG) neurons and across human Nav1.1, 1.6, 1.7, and 1.8 subtypes, with mutagenesis confirming the conserved intracellular binding pocket. In an osteoarthritis mouse model, local intra‑articular ND‑Tam rapidly reduces knee hyperalgesia with efficacy comparable to, or better than, clinically used agents such as cannabidiol and lidocaine at similar or lower doses. Collectively, the data support a differentiated, non‑opioid analgesic technology platform using tamoxifen‑like chemistry optimized for Nav targeting that offers a promising alternative to current pain therapies.
This invention introduces tamoxifen‑derived small molecules, led by N‑desmethyl tamoxifen (ND‑Tam), that bind a newly defined “tamoxifen receptor” site inside voltage‑gated sodium channels in sensory neurons as novel candidates for potent, state‑dependent pain relief. By blocking voltage-gated sodium channels (Nav) more strongly when they are active or inactivated at depolarized potentials, these compounds preferentially silence hyperexcitable nociceptors while sparing normally firing neurons. The inventors demonstrate robust inhibition of sodium currents and action potential firing in vitro in dorsal root ganglion (DRG) neurons and across human Nav1.1, 1.6, 1.7, and 1.8 subtypes, with mutagenesis confirming the conserved intracellular binding pocket. In an osteoarthritis mouse model, local intra‑articular ND‑Tam rapidly reduces knee hyperalgesia with efficacy comparable to, or better than, clinically used agents such as cannabidiol and lidocaine at similar or lower doses. Collectively, the data support a differentiated, non‑opioid analgesic technology platform using tamoxifen‑like chemistry optimized for Nav targeting that offers a promising alternative to current pain therapies.
APPLICATIONS
- Non‑opioid pain therapeutics for treatment of nociceptive, inflammatory, and neuropathic pain
- Peripherally targeted joint and regional pain relief via local (e.g., intra‑articular, peri‑nerve, topical) administration for osteoarthritis, post‑surgical pain, and focal neuropathic pain
- Disease‑specific chronic pain management: Adjunct or alternative therapy where Nav hyperexcitability is implicated
- Dysfunctional pain treatment: Offers a potential solution for pain conditions with unclear origins.
- Tamoxifen‑analogue optimization platform and Tool compounds for dissecting Nav biology
ADVANTAGES
-
Novel Nav binding site and mechanism targets a unique intracellular “tamoxifen receptor” pocket in Nav channels with a dual “bind‑and‑plug” mechanism distinct from traditional sodium channel blockers.
-
State‑ and voltage‑dependent block of hyperexcitable neurons: strong preference for open/inactivated states and more depolarized holding potentials, enhancing block of overactive nociceptors while sparing resting neurons
-
Higher functional potency and superior efficacy vs. standard comparators: ND‑Tam outperforms carbamazepine and cannabidiol in inhibiting Nav currents and DRG firing in vitro, and matches or exceeds lidocaine and CBD in a joint pain mouse model at much lower molar doses
-
Broad-spectrum relief: Targets multiple pain mechanisms, including nociceptive, inflammatory, and neuropathic pain.
- Rapid action: Demonstrates quick onset of pain relief in preclinical models.
- Potential safety benefits: May lower side effects compared to traditional pain medications.
- Repurposing leverage and reduced hormonal liability: Builds on extensive human safety and PK data for tamoxifen, while focusing on metabolites/analogs like ND‑Tam with greatly reduced estrogen receptor affinity
-
Modality and delivery flexibility: Chemistry amenable to systemic oral agents or locally delivered formulations for joint or regional pain, with clear structure–activity relationships guiding design of next‑generation Nav‑selective analgesics
PUBLICATIONS
 KEYWORDS
KEYWORDS
Neurology, pain management, neuropathic pain, nociceptive pain, inflammatory pain, tamoxifen, non-opioid, Nav, voltage-gated sodium channels, sodium channel blocker, small molecule, targeted therapy, therapeutics
Patent Information:
| Title |
App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Expire Date |
Categories:
Life Sciences > Therapeutics
Life Sciences > Biomarkers & Biomedical Research Tools
Keywords:
Chronic pain
Neurology
Non-opioid
Pain management
Small molecule
Therapeutics