SHORT DESCRIPTION
For protein engineering and biomaterial synthesis, this technology programs crystal structures by precisely positioning DNA ligands, offering enhanced control over crystal architecture.
INVENTORS
- Chad Mirkin*, PhD
-
George B. Rathmann Professor of Chemistry, Northwestern University Weinberg College of Arts and Sciences
-
Professor of Medicine - Hematology/Oncology, Northwestern University Feinberg School of Medicine
-
Professor of Biomedical Engineering, Chemical & Biological Engineering and Materials Science & Engineering, Northwestern University McCormick School of Engineering
-
Director of the International Institute for Nanotechnology (IIN)
- Janet McMillan
- Oliver Hayes
- Peter Winegar
* Principal Investigator |
NU Tech ID: NU 2018-204
IP STATUS
US Patent Application #17/311,108 Pending (view patent details)
DEVELOPMENT STAGE
TRL-3 Experimental Proof-of-Concept: Active R&D in the lab has validated the feasibility of programmable protein crystallization.
|
BACKGROUND
Current protein crystallization methods lack precise control over crystal structures. Conventional approaches rely on unpredictable protein-protein interactions and yield inconsistent crystal architectures. This shortfall hinders progress in applications such as catalysis, structural determination, and advanced biomaterials fabrication.
ABSTRACT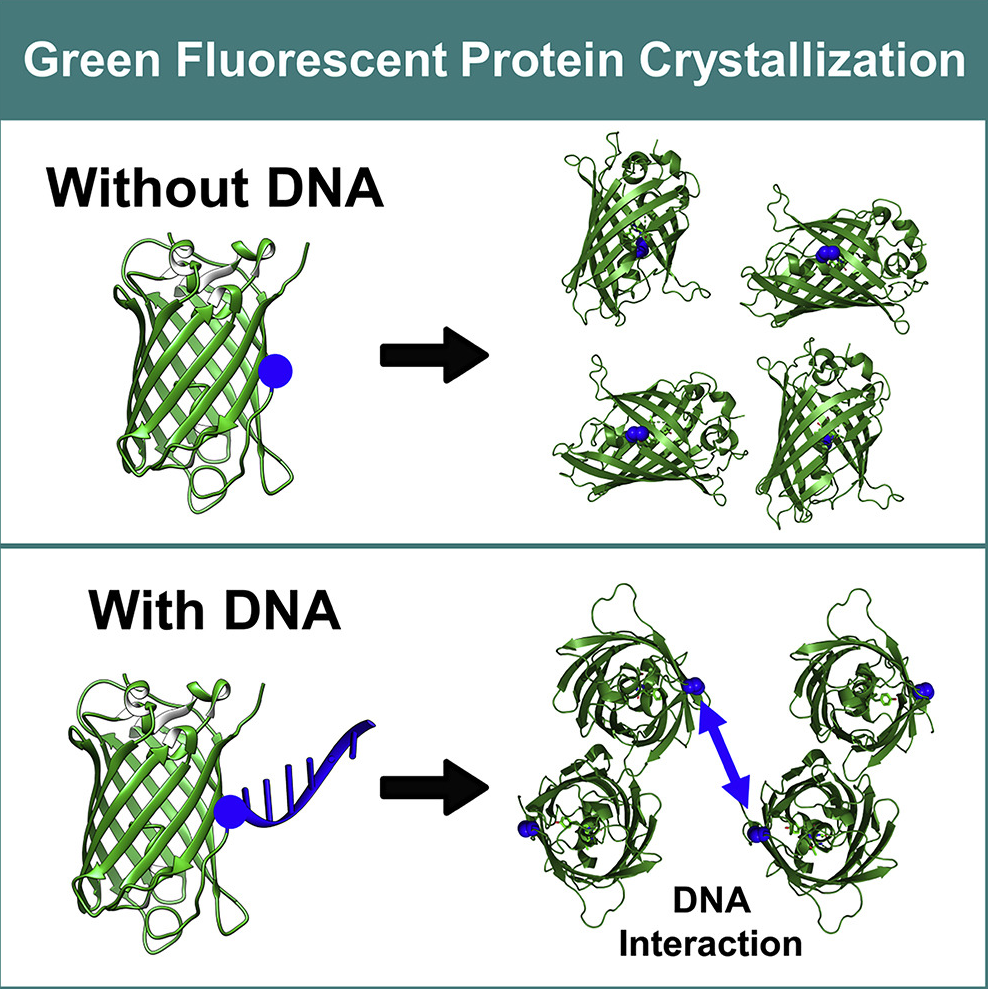
This invention programs protein crystal structure by conjugating DNA ligands to defined sites on protein surfaces. It directs crystallization via both DNA hybridization and native protein-protein interactions. The method enables fine-tuning of symmetry, topology, porosity, and reactive site orientation. Experimental results demonstrate that variations in DNA sequence and placement modulate the crystal structure. This programmable approach establishes a new strategy for designing functional protein materials.
APPLICATIONS
- Protein structural determination: Enables high-resolution analysis of protein conformations.
- Synthesis of multi-component protein crystals: Facilitates the creation of crystals with tunable architecture.
- Cascade biocatalysis: Supports design of complex catalytic pathways within a single crystal.
- Enantiomeric separations: Provides a platform for precision separations using designed crystal environments.
ADVANTAGES
- Programmable structure control: Precisely tunes symmetry, topology, and porosity for targeted applications.
- Modular design: Leverages designable DNA ligands to direct crystallization independently of protein identity.
- Enhanced reproducibility: Achieves consistent crystal assembly with predictable performance.
- Versatile functionality: Supports applications spanning catalysis, structural determination, and separation processes.
PUBLICATIONS